The House Center for Auditory Prosthesis is focused on the innovation of hearing devices including hearing aids, cochlear implants, auditory brainstem implants, and new devices.
About Auditory Prostheses
Over 430 million people worldwide suffer from debilitating hearing loss. For people with mild to moderate hearing loss, hearing aids can amplify the acoustic signal into the audible range. For people with severe-to-profound hearing loss, cochlear implants can restore hearing by electrically stimulating the surviving auditory nerves in the cochlea. For people who cannot benefit from hearing aids or cochlear implants, an auditory brainstem implant can restore hearing by stimulating higher levels of the auditory system (e.g., the cochlear nucleus and auditory brainstem).
Each of these devices provides different amounts of sound information and sound quality. Hearing aids transmit low-to-mid frequency sounds well, but not high frequency sounds. Cochlear implants can transmit most frequencies, but the sound quality is much poorer than with acoustic hearing.
Increasing numbers of cochlear implant patients have usable acoustic hearing in the implanted or non-implanted ear. Combining acoustic hearing with a cochlear implant often improves sound perception, but much work remains to optimize cochlear implants to work well with acoustic hearing. Much of our research is aimed at improving perception with combined acoustic and electric hearing. Hearing aid signal processing may also need to be optimized to work with cochlear implants. The optimization of cochlear implant and hearing aid signal processing for combined acoustic and electric hearing may also benefit patients who only use a cochlear implant or a hearing aid.
Areas of Research
House Institute has long been at the forefront of research and development for hearing aids, cochlear implants and auditory brainstem implants. Our research is focused on improving patient outcomes for complex listening demands such as binaural listening, speech understanding in challenging environments, and music perception. We are committed to improving performance with auditory prostheses, especially for combined acoustic and electrical hearing.
Personalized Optimization of Hearing Devices
Patients may differ in terms of hearing devices, hearing status, and listening environments. Rather than a “one-size-fits-all” approach, we are researching ways to optimize hearing aids, cochlear implants, and auditory brainstem implants to address individual patient needs. This research includes patient-driven optimization of hearing devices for different listening demands, as well as adjustment of device settings for better integration of devices, such as bilateral hearing aids, bilateral cochlear implants, and combined use of hearing aids and cochlear implants.
Patient-Specific Factors That Affect Outcomes
There remains great variability in outcomes for hearing aids, cochlear implants, and auditory brainstem implants. Some of this variability may be due to patient-specific factors such as the amount of residual acoustic hearing, etiology of deafness, age at onset of deafness, duration of deafness, electrode position in the cochlear, and patterns of neural health in the cochlea, etc. Greater understanding of these factors may help to better predict implant outcomes, and indicate which intervention is best and when it should be performed. The House Institute provides a vast resource with which to research these factors. We are exploring novel approaches towards understanding patient-specific patterns, such as proteomic, metabolic, and genomic analyses of tissue samples and behavioral measures of neural health.
Optimization of Clinical Fitting for Auditory Brainstem Implants
Clinical fitting for patients with auditory brainstem implants is especially challenging, as the pitch organization across electrodes is unclear and generally disordered. It is also important to activate electrodes that provide only auditory sensation, rather than non-auditory side effects. We are exploring ways to use objective measures collected during surgery (e.g., auditory brainstem responses) to predict which electrodes should be activated during clinical fitting.
Research Goals
Patient-Driven Optimization
of Signal Processing
Advances in mobile technology increasingly allow for greater patient control to optimize signal processing of auditory prostheses for different listening conditions. With artificial intelligence (AI), much of this optimization can occur automatically. However, it is important to know how to optimize the signal processing for different listening demands and environments. We will explore the possibilities of patient-driven optimization for various listening contexts, as well as automatic optimizations via cloud computing and AI.
Improving Clinical Fitting for Patients with Usable Acoustic Hearing
There is limited guidance regarding clinical fitting for patients with useable acoustic hearing in the implanted or non-implanted ear. It is important to balance loudness levels between acoustic and electric hearing with and across ears to maximize performance, especially for binaural perception. We will explore the effects of loudness imbalance between acoustic and electric hearing in terms of speech performance, sound quality, localization, and fusion. This research will help to develop hardware and software solutions for both clinical fitting and research.
Improved Music Perception
While the cochlear implant often provides good speech understanding, music perception and sound quality is much poorer. Combined acoustic and electric hearing is currently the best approach to improve music perception, but optimization of the cochlear implant may be very different for music than for speech. We will initially explore music perception in single-sided deaf cochlear implant patients, who have normal acoustic hearing in one ear and a cochlear implant in the other ear. This unique patient population can provide important within-patient comparisons between acoustic and electric hearing. This information will inform optimization of electric hearing in the context of usable acoustic hearing.
Emerging Technology
At present, the major bottleneck for cochlear implants is the limited spectral resolution, which typically allows for far fewer spectral channels than the number of implanted electrodes. New electrode designs are needed to improve spectral resolution. Reducing trauma during surgery may also help to preserve the health of auditory nerves. We aim to develop, evaluate, and implement new cochlear implant technologies to improve outcomes.
Previous Contributions to Auditory Prosthesis Science
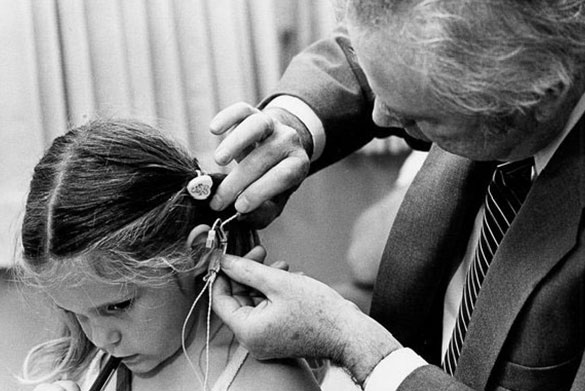
- World’s first cochlear implant, designed by William House, MD, and implanted in 1961
- First cochlear implant for clinical trials, designed by William House, MD, and Jack Urban in the 1970s
- Development of a single-electrode cochlear implant prototype that went on to become the first commercially available cochlear implant
- Development and implantation of the world’s first auditory brainstem implant in 1979
- First pediatric cochlear implant in 1981
- Development of the House/3M cochlear implant device in 1984, the first ever FDA approved implant
- Development of research interfaces that allow for direct control of cochlear implant simulation
- First digital hearing aid in the 1990s
- Developed and performed clinical trial of the PABI (Penetrating Auditory Brainstem Implant) in the early 2000s
- Part of the research effort to obtain FDA approval for cochlear implants in patients with single-sided deafness in the 2010s
- Definitive study on electric stimulation safety limits for nerves throughout the body













